Engineers Have Grown A Major Piece of The Human Heart In Miniature, And It Beats
This is incredible.
By: Mike McRae | Science Alert
Though research into treatments for cardiovascular disease has come a long way in recent decades, heart problems still claim the lives of nearly 18 million people around the world each year.
A tiny working model of a human ventricle could open fresh new ground in developing novel drugs and therapies, and for studying the development of cardiovascular conditions, giving researchers an ethical, more accurate alternative to existing approaches.
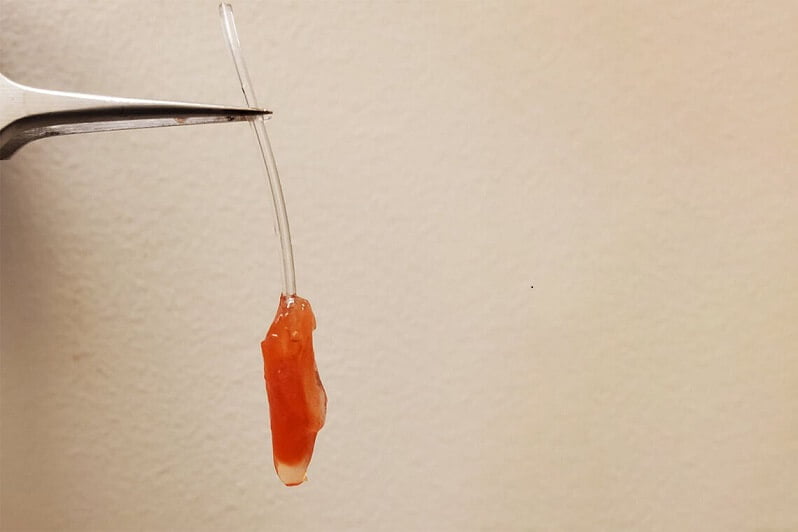
Researchers from the University of Toronto and University of Montreal in Canada reverse-engineered a millimetre-long (0.04 inches) vessel that not only beats like the real deal, but pumps fluid just like the muscular exit-chamber of a human embryo’s heart.
“With our model, we can measure ejection volume – how much fluid gets pushed out each time the ventricle contracts – as well as the pressure of that fluid,” says University of Toronto biomedical engineer, Sargol Okhovatian.
“Both of these were nearly impossible to get with previous models.”
There are typically just a handful of options for studying the ways a diseased or healthy heart channels blood.
Organs that are no longer fully functional, such as those removed in an autopsy, provide authenticity without the activity. Tissue cultures might provide a window into biochemical functionality, but they don’t fully capture the hydraulics of a three-dimensional, pulsing mass.
An animal model allows researchers to test how a living heart functions as a pump under the influence of newly developed treatments, but isn’t always the most ethical option.
Joining a wave of 3D models of body parts that develop and behave just as nature intended (without unfolding into fully functional organs), this new heart-like organ was grown in a lab using a mix of synthetic and biological materials.
The cells themselves were derived from the cardiovascular tissues of young rats, and then grown on a layer of scaffold printed out of a polymer with grooves for directing the tissue’s growth.
This flat mesh forced the structure to mimic the alignment of heart muscle fibres of a human left ventricle – the bulky final chamber that launches blood into the aorta with one mighty squeeze.
To turn the triple-layered stack of heart cells into something that more resembles a pulsing chamber, the team used a cone-shaped shaft they dubbed a mandrel. A quick roll in the tissue sample, and presto – a simple ventricle. All that was required to make this itty-bitty tube of cardiac muscle cells beat was a series of small electrical shocks.
“Until now, there have only been a handful of attempts to create a truly 3D model of a ventricle, as opposed to flat sheets of heart tissue,” says senior author Milica Radisic, a chemist from the University of Toronto.
“Virtually all of those have been made with a single layer of cells. But a real heart has many layers, and the cells in each layer are oriented at different angles. When the heart beats, these layers not only contract, they also twist, a bit like how you twist a towel to wring water out of it. This enables the heart to pump more blood than it otherwise would.”
With an internal diameter of just half a millimetre (0.02 inches), the vessel barely manages to eject liquid at a pressure of around 5% of an adult’s heart.
Still, the model is a great proof of concept, one that could in time be bulked up to include more tissue layers to represent a stronger system.
It’s even possible that with time the scaffold could be removed and a medley of human-derived tissues could be incorporated, not just improving the structure as a model but leading the way to a fully functional, transplantable organ.
“With these models, we can study not only cell function, but tissue function and organ function, all without the need for invasive surgery or animal experimentation,” says Radisic.
“We can also use them to screen large libraries of drug candidate molecules for positive or negative effects.”
This research was published in Advanced Biology.
This article was originally published by Science Alert.
